A Quality Management System is a structured framework that businesses use to standardize processes, monitor quality, and consistently meet customer requirements. However, many businesses still struggle with recurring defects, customer complaints, and inconsistent operational results across departments.
Additionally, the growing reliance on digital quality tools reflects rising global demand for structured systems. According to a market report by Grand View Research, the quality management software market was valued at USD 12.26 billion in 2025 and is projected to reach USD 28.82 billion by 2033, growing at an 11.5% CAGR.
Moreover, customers today expect consistent quality across every interaction, whether in products or services. Therefore, companies that fail to maintain reliable quality controls risk losing loyal customers to competitors who deliver faster, safer, and more dependable results.
So, in this article, we will explore the fundamentals of quality management systems, including their principles, tools, and benefits, and also review the leading QMS software solutions that help organizations maintain compliance and improve long-term operational performance.
- A Quality Management System is a structured policy, processes, and responsibilities that help organizations maintain consistent quality standards across operational activities.
- A Quality Management System is needed by companies to maintain consistency, meet regulatory requirements, reduce operational risks, and improve customer satisfaction outcomes.
- Quality Management System Tools and Techniques enable organizations to detect process gaps early and improve operational efficiency through structured monitoring practices.
- The Best QMS Software ensures organizations maintain visibility, streamline documentation workflows, and support compliance across multiple departments effectively.
- ScaleOcean QMS Software helps organizations integrate quality workflows with operations, improving traceability, efficiency, and long-term quality management performance.

What is a Quality Management System (QMS)?
A Quality Management System (QMS) is a formal system of policies, procedures, and responsibilities that organizations use to achieve quality objectives. It ensures consistent performance by standardizing workflows and aligning operational activities with clearly defined quality standards.
Additionally, a QMS helps companies document processes, track compliance, and continuously evaluate performance across departments. By maintaining structured records, companies can quickly identify inefficiencies, resolve quality control gaps, and improve operational reliability across production and service environments.
Most modern organizations integrate their QMS into digital platforms that enable real-time monitoring and centralized data management. Consequently, managers gain immediate access to performance metrics, allowing faster decision-making while reducing delays caused by manual reporting or scattered documentation.
Furthermore, a well-implemented QMS promotes accountability among employees by clearly defining responsibilities and expected outcomes. When every team member understands their role in maintaining quality, organizations reduce confusion, minimize defects, and strengthen collaboration across functional units.
Why Do Companies Need A QMS?
Companies need a Quality Management System because maintenance management is crucial for consistent quality across operations, and becomes increasingly difficult as businesses scale. Without standardized procedures, teams often produce inconsistent outputs that lead to customer dissatisfaction and unnecessary operational costs.
Moreover, regulatory requirements continue to evolve across industries, requiring companies to maintain strict compliance standards. A structured QMS ensures that organizations document activities properly, maintain traceability, and demonstrate accountability during audits or regulatory inspections.
A QMS also improves operational efficiency by identifying inefficiencies and eliminating redundant processes. As teams follow standardized workflows, organizations reduce rework, optimize resource usage, and improve productivity while maintaining consistent output quality across departments.
Key Reasons Companies Implement a QMS:
- Improving process consistency: Standardized workflows reduce variation across operations, allowing teams to deliver uniform results while minimizing costly errors and production delays.
- Enhancing customer satisfaction: Reliable quality performance ensures customers receive consistent products and services, strengthening trust while increasing long-term loyalty and repeat business opportunities.
- Supporting compliance readiness: Documented procedures help organizations meet regulatory requirements efficiently, reducing the risk of penalties, failed audits, or unexpected operational disruptions.
- Driving continuous improvement: Performance tracking enables teams to analyze results, identify recurring issues, and implement targeted improvements that enhance efficiency and overall product reliability.
Types of Quality Management Systems
Organizations apply different types of quality management systems depending on their industry, operational risks, and regulatory requirements. Therefore, selecting the appropriate system helps companies align quality practices with compliance standards while maintaining consistent operational performance across departments.
Additionally, standardized management systems provide structured frameworks that guide documentation, risk control, and performance monitoring. By following recognized standards, businesses strengthen process reliability, improve accountability, and ensure that teams follow consistent quality procedures throughout daily operations.
Many industries rely on internationally recognized standards to maintain product safety and operational reliability. Consequently, adopting the right system allows organizations to improve transparency, reduce compliance risks, and demonstrate commitment to quality during audits and regulatory assessments.
Understanding the key differences between major quality management software system standards helps organizations select the most suitable framework. Each system focuses on specific operational priorities, ensuring that businesses address industry-specific risks while maintaining long-term quality and safety performance.
ISO 9001:2015 (Quality Management Systems)
ISO 9001:2015 is one of the most widely adopted quality management standards across industries worldwide. It focuses on establishing consistent processes, improving customer satisfaction, and ensuring that organizations meet regulatory requirements through structured documentation and continuous performance evaluation.
Additionally, this standard emphasizes risk-based thinking, encouraging organizations to identify potential quality issues before they escalate into major problems. By applying preventive actions and continuous monitoring, companies improve operational stability while reducing costly errors and customer complaints.
ISO 13485:2016 (Medical Devices Regulatory & Quality Management)
ISO 13485:2016 focuses specifically on quality management software system requirements for organizations involved in medical device production. It ensures that companies consistently design, manufacture, and deliver medical products that meet strict regulatory and safety requirements across global healthcare markets.
Moreover, this standard emphasizes product traceability and risk management throughout the product lifecycle. By documenting every stage of development and distribution, companies reduce safety risks while ensuring that medical devices remain reliable and compliant with international health regulations.
ISO 14001 (Environmental Management Systems)
ISO 14001 focuses on managing environmental responsibilities while maintaining operational efficiency across industries. It provides guidelines that help organizations reduce environmental impact, comply with environmental regulations, and implement sustainable practices that support long-term operational resilience.
Furthermore, this standard encourages organizations to identify environmental risks associated with their activities and develop strategies to reduce pollution. By monitoring resource usage and emissions, companies minimize waste generation while improving environmental accountability and regulatory compliance.
ISO 22000:2018 (Food Safety Management Systems)
ISO 22000:2018 addresses food safety risks across the entire food supply chain, from production to distribution. It ensures that organizations implement strict safety controls to prevent contamination while maintaining consistent product quality for consumers and regulatory compliance.
Additionally, this standard integrates hazard analysis and critical control point principles into daily operations. By identifying safety risks early, organizations maintain safe handling procedures that protect consumers while preserving product quality throughout processing and delivery stages.
ISO 22301 (Business Continuity Management Systems)
ISO 22301 focuses on maintaining operational continuity during unexpected disruptions such as natural disasters, cyberattacks, or supply chain failures. It helps organizations develop structured plans that ensure business operations continue even when unexpected risks occur.
Moreover, this standard emphasizes risk assessment and response planning to minimize operational downtime. By identifying vulnerabilities and preparing recovery strategies, companies protect critical processes while maintaining service reliability during emergencies or system failures.
ISO 45001: 2018 (Occupational Health and Safety Management Systems)
ISO 45001:2018 focuses on protecting employee health and safety across workplace environments. It provides guidelines that help organizations identify workplace hazards, reduce accident risks, and maintain safe working conditions across all operational areas.
Additionally, this standard promotes proactive risk management and employee participation in safety programs. By encouraging hazard reporting and safety awareness, organizations reduce workplace incidents while strengthening compliance with occupational health and safety regulations.
What are the 7 Quality Management Principles?
The seven quality management principles provide a structured foundation that helps organizations improve quality performance and maintain consistent operational outcomes. These principles guide businesses in aligning strategies, processes, and people toward achieving reliable and measurable results.
Additionally, these principles support organizations in strengthening internal coordination and maintaining accountability across departments. Companies can ensure employees understand responsibilities while maintaining consistent standards throughout production, service delivery, and administrative activities.
Furthermore, applying these principles strengthens organizational culture by encouraging collaboration and continuous improvement. When companies consistently follow these structured approaches, they enhance operational transparency while ensuring long-term quality performance across interconnected processes and business functions.
1. Engagement of People
Engagement of people emphasizes involving employees at every organizational level in maintaining quality standards. When individuals clearly understand their responsibilities, they contribute actively to operational success while strengthening communication, teamwork, and accountability across departments and production environments.
Moreover, organizations that encourage employee participation improve problem-solving capabilities and reduce operational disruptions. By supporting continuous learning and knowledge sharing, businesses empower employees to identify improvement opportunities while maintaining consistent quality outcomes across daily activities.
2. Customer Focus
Customer focus highlights the importance of understanding customer expectations and delivering products or services that consistently meet those needs. Organizations that prioritize customer satisfaction strengthen trust while building long-term relationships that support stable revenue growth and competitive positioning.
Furthermore, collecting customer feedback allows organizations to identify quality gaps and refine product performance effectively. By analyzing responses and responding proactively, businesses improve service reliability while ensuring their offerings remain aligned with changing customer expectations and market demands.
3. Leadership
Leadership focuses on establishing clear direction and creating an environment that supports consistent quality performance. Strong leaders communicate organizational goals clearly while ensuring employees understand expectations and remain aligned with long-term operational and strategic objectives.
Additionally, effective leadership encourages accountability and promotes a culture that values continuous improvement. When leaders actively support quality initiatives, organizations maintain consistent performance while strengthening employee motivation and ensuring resources remain aligned with operational priorities.
4. Process Approach
The process approach emphasizes managing activities as interconnected processes rather than isolated tasks. By understanding how operations interact, organizations identify inefficiencies more effectively while ensuring consistent outputs across production stages and service delivery environments.
Moreover, documenting workflows and monitoring performance allows organizations to maintain standardized operations across departments. Through structured evaluation, businesses detect process variations early while improving coordination and maintaining predictable results across operational systems.
5. Improvement
Improvement focuses on continuously enhancing products, services, and operational processes to maintain strong performance levels. Organizations that prioritize ongoing refinement will adapt more effectively to market changes while maintaining consistent quality and operational reliability.
Furthermore, improvement activities involve identifying performance gaps and implementing targeted corrective actions. By analyzing operational data regularly, organizations prevent recurring problems while strengthening long-term efficiency and maintaining alignment with evolving business objectives.
6. Evidence-Based Decision Making
Evidence-based decision-making ensures organizations rely on accurate data when evaluating performance and planning improvements. By using measurable indicators, companies reduce uncertainty while improving confidence in operational and strategic decision-making processes.
Additionally, consistent data collection allows organizations to identify performance trends and predict potential risks early. Through structured reporting systems, businesses strengthen transparency while ensuring decisions remain supported by reliable and verifiable operational information.
7. Relationship Management
Relationship management focuses on maintaining strong partnerships with suppliers, customers, and stakeholders who influence operational outcomes. Organizations that build reliable relationships improve coordination while ensuring consistent material quality and dependable service performance.
Moreover, maintaining open communication with stakeholders helps organizations respond quickly to operational challenges. By strengthening collaboration and monitoring supplier performance, businesses reduce disruptions while ensuring stable operations across supply chains and interconnected service networks.
Why is a Quality Management System Important?
A Quality Management System (QMS) plays a critical role in helping organizations maintain consistent quality across operations. Without a structured framework, teams often overlook process gaps, leading to recurring defects, customer dissatisfaction, and rising operational costs that weaken long-term business stability.
Moreover, businesses face increasing pressure to deliver reliable results while meeting strict regulatory expectations. Therefore, implementing a structured QMS allows organizations to improve transparency, strengthen accountability, and maintain operational control across production, service delivery, and administrative functions.
- Customer Satisfaction: A structured QMS ensures products and services consistently meet defined standards, reducing customer complaints while strengthening trust, improving brand credibility, and encouraging repeat purchases that support long-term business growth across competitive markets.
- Regulatory Compliance: Standardized documentation and controlled workflows help organizations meet industry regulations efficiently, reducing risks of penalties, failed inspections, and unexpected disruptions caused by incomplete records or inconsistent compliance procedures.
- Efficiency and Cost Reduction: Streamlined processes minimize rework, reduce material waste, and optimize resource usage, allowing organizations to control expenses while improving productivity and maintaining consistent output quality across operational departments.
- Continuous Improvement: A QMS supports regular evaluation of performance metrics, enabling organizations to identify weaknesses, implement corrective actions, and maintain steady progress toward higher efficiency and stronger long-term operational reliability.
What are the Challenges in Maintaining an Effective QMS
Maintaining an effective quality management system QMS requires continuous monitoring, clear leadership direction, and strong employee participation across departments. However, many organizations struggle to sustain system effectiveness due to operational complexity, changing requirements, and limited internal resources.
Additionally, businesses often face difficulties balancing documentation, technology adoption, and workforce readiness while maintaining compliance standards. Recognizing these challenges allows organizations to strengthen their quality frameworks while ensuring their systems remain adaptable to evolving operational demands.
- Weak Root Cause Analysis: Organizations sometimes address visible symptoms rather than investigating underlying causes, which leads to recurring quality issues that disrupt workflows and increase operational risks across production and service processes.
- Over-Documentation and Complexity: Excessive documentation creates confusion among employees, slows task execution, and increases the likelihood of inconsistent practices that reduce efficiency and weaken overall system reliability across departments.
- Static Systems in Dynamic Environments: Quality systems that fail to evolve alongside technological changes and market demands become outdated, limiting their ability to support innovation and respond effectively to new operational challenges.
- Resource Constraints: Limited budgets, insufficient staffing, and inadequate training programs reduce an organization’s ability to maintain system performance, increasing the risk of compliance failures and weakening long-term quality management effectiveness.
Quality Management System Tools & Techniques
Organizations use quality management tools to maintain consistency and monitor performance. These methods help identify inefficiencies, manage risks, and standardize workflows, improving coordination and reducing variability to ensure predictable outcomes aligned with quality standards across departments.
Moreover, combining multiple tools addresses operational challenges and improves the detection of process weaknesses. Integrating digital technologies with traditional methods boosts responsiveness and reporting accuracy, supporting continuous monitoring, compliance, and ongoing quality improvement.
Plan-Do-Check-Act (PDCA) Cycle
The Plan-Do-Check-Act cycle provides a structured framework that supports continuous process improvement through repetitive evaluation stages. Organizations begin by planning improvements, implementing changes, reviewing results, and refining strategies to maintain consistent operational performance.
Moreover, applying the PDCA cycle encourages systematic problem-solving that minimizes uncertainty during improvement initiatives. By continuously repeating the cycle, organizations strengthen operational control while ensuring that improvements remain measurable, sustainable, and aligned with long-term quality objectives.
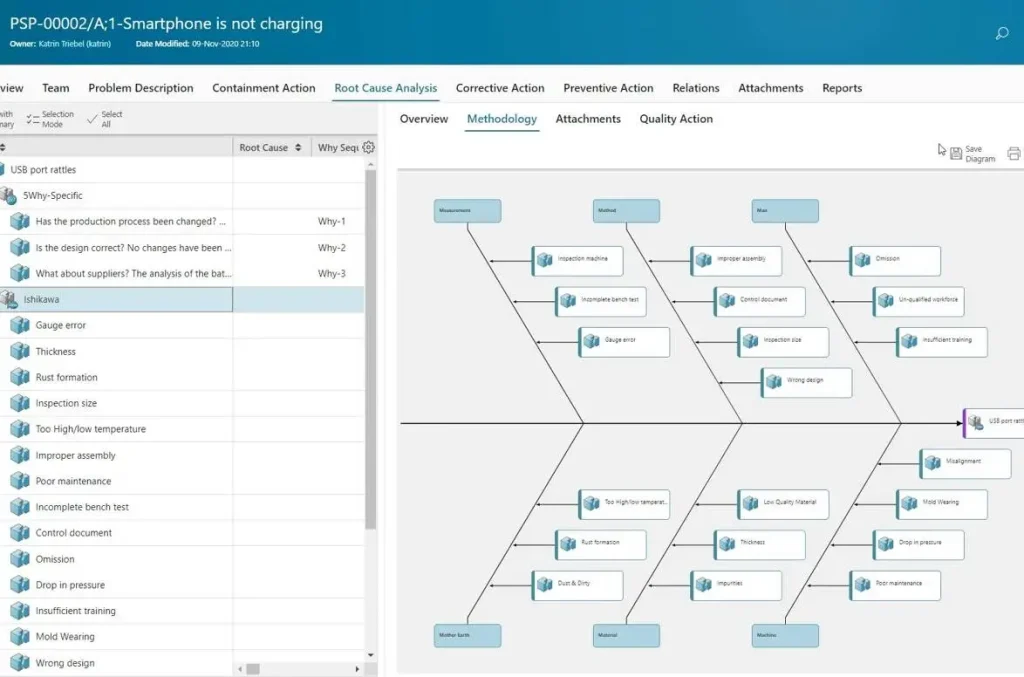
Cause-and-Effect (Fishbone) Diagram
The cause-and-effect diagram, commonly known as the fishbone diagram, helps organizations identify potential root causes of operational problems. By categorizing contributing factors visually, teams analyze relationships between variables while improving accuracy in diagnosing recurring quality issues.
Additionally, this technique supports collaborative problem-solving by encouraging team discussions around possible causes of defects or inefficiencies. When organizations apply structured root cause analysis methods, they improve decision accuracy while preventing repeated failures that affect operational reliability.
Six Sigma
Six Sigma focuses on reducing process variation and improving product consistency through data-driven methodologies. Organizations applying Six Sigma analyze defects statistically, allowing teams to measure performance precisely while implementing targeted improvements that reduce errors across production cycles.
Furthermore, this technique promotes disciplined project management that strengthens accountability and encourages continuous measurement of operational outcomes. By applying structured improvement phases, organizations enhance productivity across complex operational environments.
Lean Manufacturing
Lean manufacturing emphasizes eliminating waste and maximizing value across operational processes. Organizations applying lean principles identify unnecessary activities, reduce excess inventory, and streamline workflows to maintain efficiency while ensuring consistent output quality across production stages.
Moreover, lean methods encourage organizations to focus on customer value while optimizing resource usage. By removing non-essential steps, teams improve productivity while maintaining strong coordination between departments responsible for delivering reliable and timely results.
Just-in-Time (JIT) Manufacturing
Just-in-Time manufacturing focuses on producing and delivering materials only when needed to reduce storage requirements and minimize inventory waste. Organizations applying this technique improve production timing while maintaining flexibility across dynamic operational schedules.
Additionally, JIT strengthens supply chain coordination by ensuring that materials arrive exactly when required. By aligning supplier schedules with production demands, organizations maintain efficiency while reducing storage costs and minimizing risks associated with excess inventory accumulation.
Quality Management System Software (QMS)
Quality Management System software supports organizations in managing quality processes through centralized digital platforms that improve coordination and documentation accuracy. By automating data collection and workflow tracking, businesses reduce manual errors while strengthening visibility across activities.
Implementing dedicated quality management software allows real-time performance monitoring and structured compliance documentation. Solutions like ScaleOcean QMS software integrate quality management to standardize workflows, improve reporting, and ensure consistent quality in changing environments.
As organizations adopt digital tools to strengthen quality control, selecting the best quality management software becomes a critical step. Therefore, understanding the leading QMS providers will help businesses identify solutions that align with operational complexity, compliance needs, and long-term improvement strategies.
Top 8 QMS Software Solutions & Providers
Selecting the right quality management software plays an essential role in maintaining consistent quality performance across operational activities. As organizations transition from manual processes to digital platforms, they require reliable tools that support compliance, monitoring, and structured quality documentation.
Moreover, evaluating multiple quality management software system providers helps organizations compare functionality, scalability, and system flexibility before making implementation decisions. By understanding available solutions, businesses identify platforms that align with operational complexity while supporting long-term quality management.
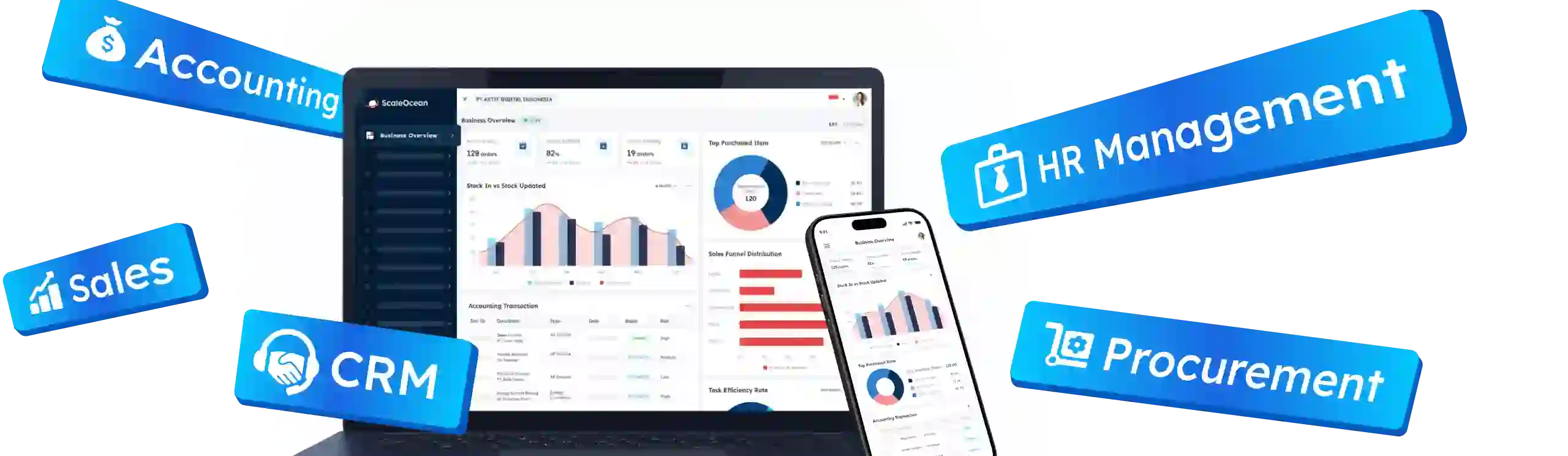
1. ScaleOcean
ScaleOcean QMS Software is designed to manage compliance, continuous improvement, and operational integration within a centralized platform. It allows organizations to maintain standardized quality workflows while coordinating activities across departments through one connected digital environment.
Moreover, the system integrates directly with production, inventory, purchasing, and distribution processes, enabling organizations to trace quality issues from raw materials through manufacturing stages until final delivery. This end-to-end traceability strengthens visibility and supports faster root cause identification and corrective action execution.
Additionally, ScaleOcean provides full customization capabilities that allow organizations to configure workflows according to internal standard operating procedures. Companies can tailor inspection forms, approval flows, and documentation processes, ensuring the system aligns with industry requirements and diverse operational practices.
The platform also supports unlimited user access with flat pricing based on system complexity rather than user volume. Furthermore, it is optimized for Singapore tax compliance and reporting standards, enabling eligibility for digitalization grants up to 50-70% that support structured quality transformation initiatives.
Key Features of ScaleOcean QMS Software:
- Document Control: Manages complete document lifecycles digitally, including version control, approval workflows, and secure storage, helping organizations maintain structured documentation that supports strict regulatory audits and consistent operational transparency.
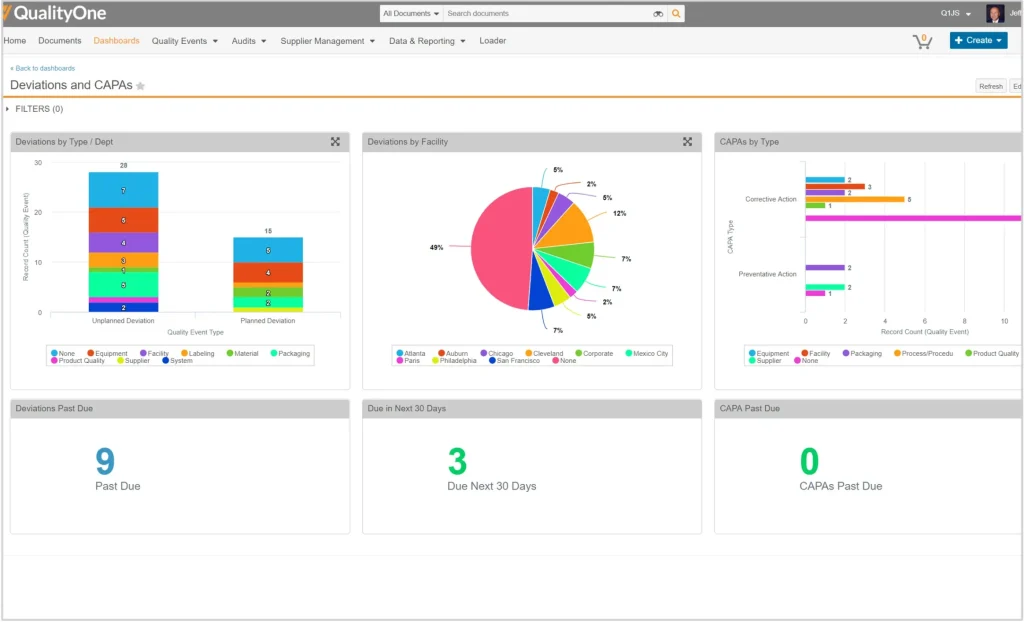
- CAPA Management: Supports root cause analysis and structured corrective workflows, enabling organizations to track preventive and corrective actions from identification to closure while reducing risks associated with unresolved quality incidents.
- Non-Conformance Management (NCR): Records defects and classifies issues based on severity levels, allowing teams to monitor investigation progress while ensuring corrective measures remain documented and traceable across quality processes.
- Audit Management: Facilitates internal and external audit preparation through scheduling tools, structured checklists, and findings documentation, helping organizations maintain audit readiness while improving follow-up accountability across departments.
- Advanced IoT & Real-Time Monitoring: Integrates with machines and sensors to monitor operational parameters automatically, enabling predictive quality control that reduces downtime risks and strengthens early detection of production deviations.
| Pros | Cons |
|---|---|
|
|
Best for: suited for mid-sized and large enterprises that require integrated quality visibility across production, inventory, and distribution processes, especially organizations operating in regulated industries that demand strong traceability, compliance readiness, and customizable workflow control across departments.
2. Propel Software
Propel QMS software is designed to support product quality management across product development and manufacturing environments. It helps organizations maintain structured quality workflows while ensuring alignment between product lifecycle activities and regulatory documentation requirements.
Additionally, the platform supports cloud-based operations that enable collaboration between distributed teams and departments. By centralizing product and quality data, organizations improve coordination while maintaining consistent traceability across product development and quality validation stages.
Key Features:
- Product lifecycle quality management
- Change and document management
- Supplier quality tracking
- Cloud-based collaboration tools
| Pros | Cons |
|---|---|
|
|
Best for: Mid-sized to large manufacturing or product-based companies that manage product lifecycle quality across multiple departments, especially those requiring collaboration between engineering, quality, and regulatory teams.
3. Veeva Vault QMS
Veeva Vault QMS software is designed to support quality and compliance management within highly regulated industries. It helps organizations maintain controlled documentation processes while supporting structured regulatory readiness across operational and quality management environments.
Moreover, the platform supports centralized workforce readiness by linking training records with compliance workflows. By maintaining structured documentation, organizations improve audit preparedness while ensuring consistent alignment with regulatory and quality standards.
Key Features:
- Quality document control
- Training and compliance tracking
- Change management workflows
- Audit preparation tools
| Pros | Cons |
|---|---|
|
|
Best for: Large enterprises in regulated industries such as pharmaceuticals, biotechnology, and life sciences that require strict documentation control, regulatory alignment, and structured audit preparation workflows.
4. Plex QMS
Plex QMS software is designed to support quality management within production-driven manufacturing environments. It helps organizations maintain standardized workflows while monitoring quality performance across manufacturing operations and production-related processes.
Additionally, the platform connects operational workflows with structured quality tracking activities. By linking production performance with quality monitoring, organizations improve responsiveness while maintaining consistent visibility across factory and operational environments.
Key Features:
- Manufacturing quality monitoring
- Production-linked quality tracking
- Compliance reporting tools
- Process performance monitoring
Supports coordination between production and quality teams
|
|
Best for: Mid-sized to large manufacturing companies that operate production-driven environments and require consistent monitoring of quality performance across multiple production lines or facilities.
5. Effivity
Effivity QMS software is designed to support organizations in managing compliance and quality documentation within structured operational environments. It helps businesses maintain standardized workflows while ensuring documentation remains organized across departments and regulatory processes.
Additionally, the platform supports centralized tracking of quality activities, allowing teams to maintain visibility across inspections, audits, and compliance reporting tasks. By simplifying documentation workflows, organizations improve consistency while reducing manual tracking across quality operations.
Key Features:
- Compliance documentation management
- Audit tracking tools
- Risk and incident reporting
- Workflow automation support
| Pros | Cons |
|---|---|
|
|
Best for: Small to mid-sized organizations in regulated industries that require structured compliance documentation, standardized reporting workflows, and centralized audit tracking without highly complex system customization requirements.
6. Siemens QMS
Siemens QMS software is designed to support large-scale industrial environments that require structured quality management across complex operations. It helps organizations maintain consistency across engineering, production, and operational workflows within highly coordinated industrial settings.
Moreover, the platform integrates quality activities with operational systems, allowing teams to maintain consistent visibility across production quality performance. This integration supports long-term quality monitoring across complex industrial and manufacturing environments.
Key Features:
- Enterprise-level quality management tools
- Engineering and production integration
- Quality performance monitoring
- Industrial process documentation
| Pros | Cons |
|---|---|
|
|
Best for: Large manufacturing and industrial enterprises managing complex production environments that require coordinated engineering, production, and quality management across multiple operational facilities.
7. Unifi
Unifi QMS software is designed to support organizations in maintaining structured quality documentation and standardized operational workflows. It helps teams manage quality-related records while ensuring consistent tracking of inspections, corrective actions, and compliance-related activities.
Additionally, the platform supports centralized quality tracking that improves transparency across operational departments. By maintaining consistent documentation processes, organizations improve traceability while reducing the risk of missing or incomplete quality records.
Key Features:
- Document control management
- Corrective action tracking
- Inspection record management
- Compliance workflow support
| Pros | Cons |
|---|---|
|
|
Best for: Small to mid-sized organizations that require structured document control, corrective action tracking, and compliance monitoring across departments with moderate operational complexity.
8. Intellect
Intellect QMS software is designed to support organizations in managing quality workflows through configurable digital processes. It enables teams to organize inspections, compliance tasks, and reporting workflows within a centralized quality management system QMS
Furthermore, the platform supports configurable workflows that allow organizations to adapt processes based on operational requirements. This flexibility helps teams maintain consistent quality tracking while supporting evolving operational demands across departments.
Key Features:
- Configurable workflow management
- Inspection and audit tracking
- Compliance reporting tools
- Risk management tracking
| Pros | Cons |
|---|---|
|
|
Best for: Mid-sized to large organizations that require configurable workflow-based quality systems to manage inspections, compliance reporting, and risk tracking across multiple departments and operational functions.
After reviewing multiple QMS providers, organizations often recognize the importance of selecting a solution that connects quality activities with broader operational workflows. ScaleOcean supports this need through an all-in-one system that integrates compliance, improvement, and operational processes within one centralized platform.
By connecting quality processes with production, inventory, and distribution workflows, ScaleOcean improves traceability while reducing delays caused by disconnected systems. To better understand how this integration supports daily operations, request a free demo of ScaleOcean to explore its capabilities.

Conclusion
A Quality Management System QMS helps organizations maintain consistent quality, improve efficiency, and meet regulatory requirements across operations. By applying structured principles, tools, and reliable software solutions, businesses strengthen process control while supporting long-term operational stability and continuous improvement.
As organizations modernize quality processes, selecting a QMS software becomes increasingly important. ScaleOcean QMS software supports it by centralizing compliance, improvement, and operational workflows, allowing teams to maintain consistent quality control while improving coordination across departments.
Integrated systems also improve traceability and reduce delays caused by disconnected workflows. Organizations seeking stronger quality control and long-term efficiency can request a free demo to explore how ScaleOcean supports structured and scalable quality management implementation.
FAQ:
1. What is a QMS checklist?
A QMS checklist is a guide used to evaluate an organization’s compliance with regulatory standards like FDA, ISO 13485, and GxP. It helps ensure that processes, documentation, and training meet compliance requirements and promote operational excellence.
2. What are the 4 types of QMS?
Common QMS approaches include continuous quality improvement (CQI), total quality management (TQM), lean manufacturing, Six Sigma, and agile QMS methodologies.
3. What are the 5 core tools of QMS?
The five core quality tools essential for manufacturing QMS are APQP, PPAP, FMEA, MSA, and SPC. These tools focus on preventing errors to achieve significant cost savings.
4. What are the three pillars of QMS?
The three pillars of a quality management system are a document management system (DMS), a learning management system (LMS), and, during clinical trials, an electronic trial master file (eTMF).